 
What are some occupations involving astronomy?.I want to finish high school in 3 years instead of 4, but I am not sure it is a good idea.When I am making a speech or a presentation in front of the class, my face or body automatically shivers.Is it still important for people who develop Web pages to know HTML? If so, why?.Do you have any tips for getting along with everybody at work? I started my first job a couple weeks ago (just for the summer).My mom and my friends say I should quit doing something, but I love all the things I do.What is your opinion of the rise of virtual actors and the fall of live ones, what do you think about virtual actors taking the place of live ones?.What do you do when you're lost when you can't concentrate and have lost your will to succeed? How can you get back on track?.How do I convince my parents to spend a few extra bucks to upgrade from a dial-up connection to broadband like a cable modem or DSL? They say I have to give some benefits for spending extra.How can I highlight my textbooks efficiently?. 
What tips can you give me for studying for a test on something I've read?.How do I choose a topic for a personal essay?.Is 8 hours of sleep really that important? I have too many projects and not enough hours in the day.What can I do if I think my teacher gave me the wrong grade?.What kind of careers are available for someone with a degree in English?.What does it mean to live in a credential society?.To study better, I want to get organized with some of the stuff I see advertised.How do I pull together all the notes I've taken to study for a test?.Is my summer vacation to Italy a good topic for my college essay? (I have pictures, too.).Will mentioning my race in my college essay increase my chances of getting in?.How do I get involved in classroom discussions without sounding stupid?.What is the easiest foreign language to learn? Which foreign language looks the best on college applications?.Is it common for people to be scared to go into high school? Can you give me some tips to survive?.How much outside class study time is recommended for every hour of class time for college freshmen?.I want to ask him on a date, but I'm not sure if I should, and if I should, how? There is a new guy at my school and I think he's cute, funny, and sweet, but he's really shy.A friend of mine told me that my favorite TV show jumped the shark." What does that even mean?".What are the best courses to take if I want to end up doing research in metaphysics?.Who was the first female Senator in the United States?.What should I consider when deciding whether to invest in a company?.How can banks afford to lend out so much money?.The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.įinding molar mass starts with units of grams per mole (g/mol). These relative weights computed from the chemical equation are sometimes called equation weights. This site explains how to find molar mass.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.Ī common request on this site is to convert grams to moles. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. 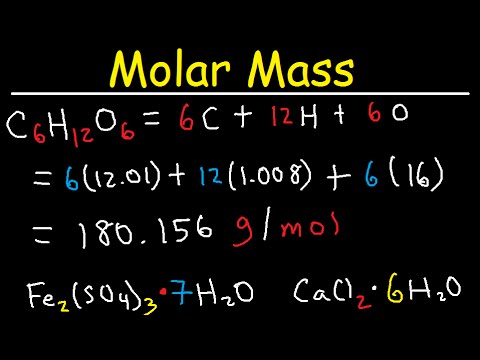
For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
